|
Some of the examples of divalent cations are Barium(Ba 2+), Calcium(Ca 2+), Magnesium(Mg 2+), etc. The cation is denoted by superscript 2+ on the symbol of atom or group of atoms. They are the basic radicals formed by the removal of hydroxide ions from the base.ĭivalent cations lose two valence electrons and attain a positive charge. Some of the examples of divalent anions are oxide (O 2-), Carbonate(CO 3 2-), etc.ĭivalent Cations are the positively charged ions having their valency (2+). An aqueous solution of 0.8 mol of cystine (100 mL) is freshly prepared and added slowly to the Cpd 0 hydrogel (0.5 g) under stirring to ensure uniform mixing. 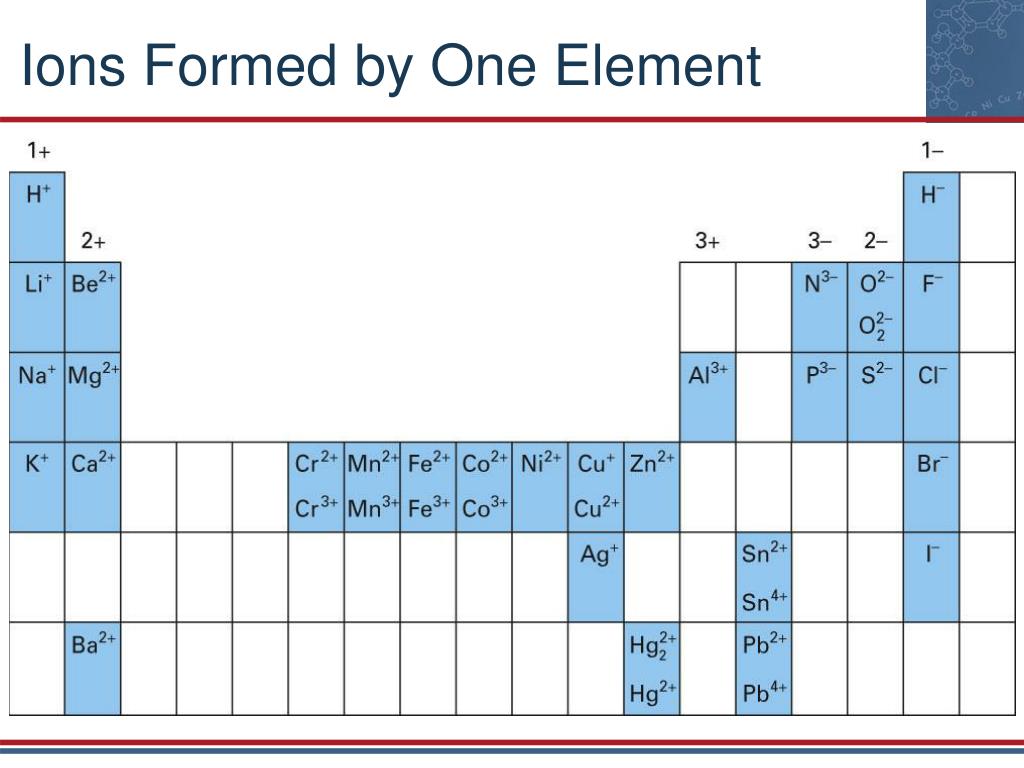
It is denoted by superscript 2- on the symbol of an atom or group of atoms. bisulfate (or hydrogen sulfate) - HSO 4. bicarbonate (or hydrogen carbonate) - HCO 3. Its good to know these ions on sight to help balance equations and predict compound formation. This means the number of electrons is higher than protons in divalent anions. Many of the common polyatomic ions have an electrical charge of -1. They are the electronegative radicals also known as bivalent anions.ĭivalent anions are formed by gaining two valence electrons and attaining a negative charge. During the formation of some compounds, atoms gain or lose electrons, and form electrically charged particles called ions (Figure 3.5.1 3.5. The cations are categorised into four types Cationsĭivalent anions are the negatively charged ions having their valency (2-). Many transition elements can generate cations as a result of electron loss. These electrons are weakly linked to the atom and can be easily withdrawn, leaving the atom with more protons than electrons, resulting in a positive-charged ion. We can see from the periodic table below that group 1 element all have one s electron in the outer orbit, whereas group 2 elements have two s electrons in the outer orbit. Because they are positively charged and migrate toward the negative electrode (cathode) in an electrical field, these ions are called “cations.” When some elements form ions, they lose one or more electrons. Software Engineering Interview Questions.Top 10 System Design Interview Questions and Answers. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
